No True Inert Placebos Were Used for Testing Most Childhood Vaccines. Why Not?
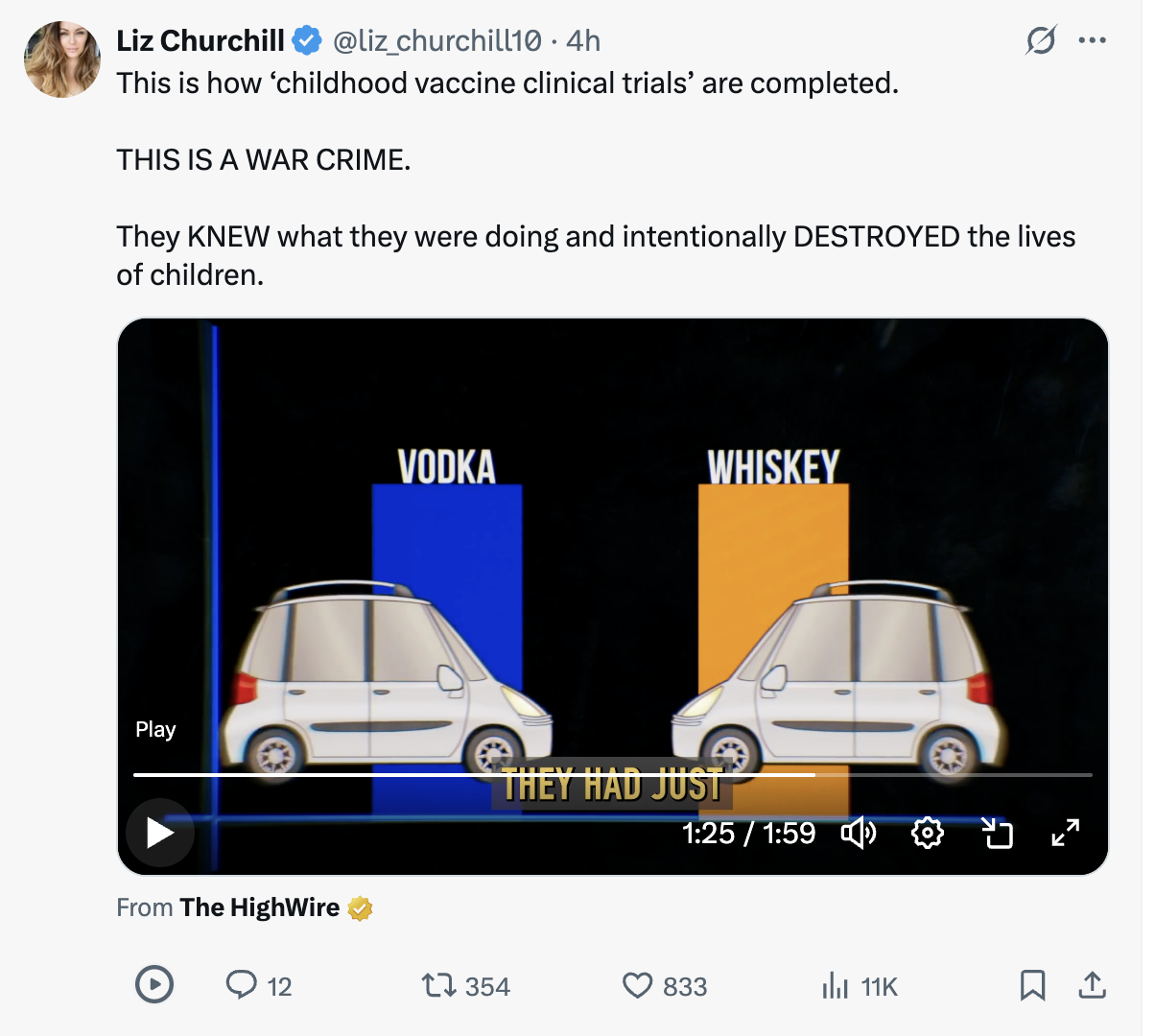
Industry allied experts claim that it would be unethical to test a vaccine against a true inert placebo like saline. Really? Del Bigtree explains that the Pharma-preferred tests (against already existing vaccines) are like testing the safety of whiskey by comparing it to the "placebo" of vodka.
But I keep coming back to the claim of "unethical." This makes no sense to me, no matter how many times I hear an industry insider make the claim. Aaron Siri explored this exact topic in his outstanding new book, Vaccines, Amen! Here's an excerpt from page 163:
Ethics of Using and Not Using a PlaceboIn response to the lack of a placebo in some trials, the FDA and vaccinologists will assert that it is unethical to evaluate the safety of an unlicensed experimental vaccine in a placebo-controlled trial when there is already a licensed vaccine for the same disease.
This ethical concern, however, rings hollow because it ethics were a real concern, the FDA would require placebo-controlled trials before licensing new experimental vaccines at least where no licensed vaccine exists to prevent the same disease. For example, the very first vaccines for chickenpox or hepatitis A, both licensed in 1995, or HPV, licensed in 2006, could have easily included a placebo control group. No ethical issue with that. But as you now know, that is not what happened because that is not how the cult of vaccinology works.
Also, consider this: conducting a placebo-controlled trial will leave a clearly defined group of children unvaccinated only for the duration of the trial in a controlled setting where they can be monitored. In contrast, injecting a vaccine into millions of children in an uncontrolled setting without first assessing its safety in a placebo-controlled trial is, to any objective, reasonable observer, grossly unethical conduct."