Industry allied experts claim that it would be unethical to test a vaccine against a true inert placebo like saline. Really? Del Bigtree explains that the Pharma-preferred tests (against already existing vaccines) are like testing the safety of whiskey by comparing it to the “placebo” of vodka.
But I keep coming back to the claim of “unethical.” This makes no sense to me, no matter how many times I hear an industry insider make the claim. Aaron Siri explored this exact topic in his outstanding new book, Vaccines, Amen! Here’s an excerpt from page 163:
Ethics of Using and Not Using a Placebo
In response to the lack of a placebo in some trials, the FDA and vaccinologists will assert that it is unethical to evaluate the safety of an unlicensed experimental vaccine in a placebo-controlled trial when there is already a licensed vaccine for the same disease.
This ethical concern, however, rings hollow because it ethics were a real concern, the FDA would require placebo-controlled trials before licensing new experimental vaccines at least where no licensed vaccine exists to prevent the same disease. For example, the very first vaccines for chickenpox or hepatitis A, both licensed in 1995, or HPV, licensed in 2006, could have easily included a placebo control group. No ethical issue with that. But as you now know, that is not what happened because that is not how the cult of vaccinology works.
Also, consider this: conducting a placebo-controlled trial will leave a clearly defined group of children unvaccinated only for the duration of the trial in a controlled setting where they can be monitored. In contrast, injecting a vaccine into millions of children in an uncontrolled setting without first assessing its safety in a placebo-controlled trial is, to any objective, reasonable observer, grossly unethical conduct.”

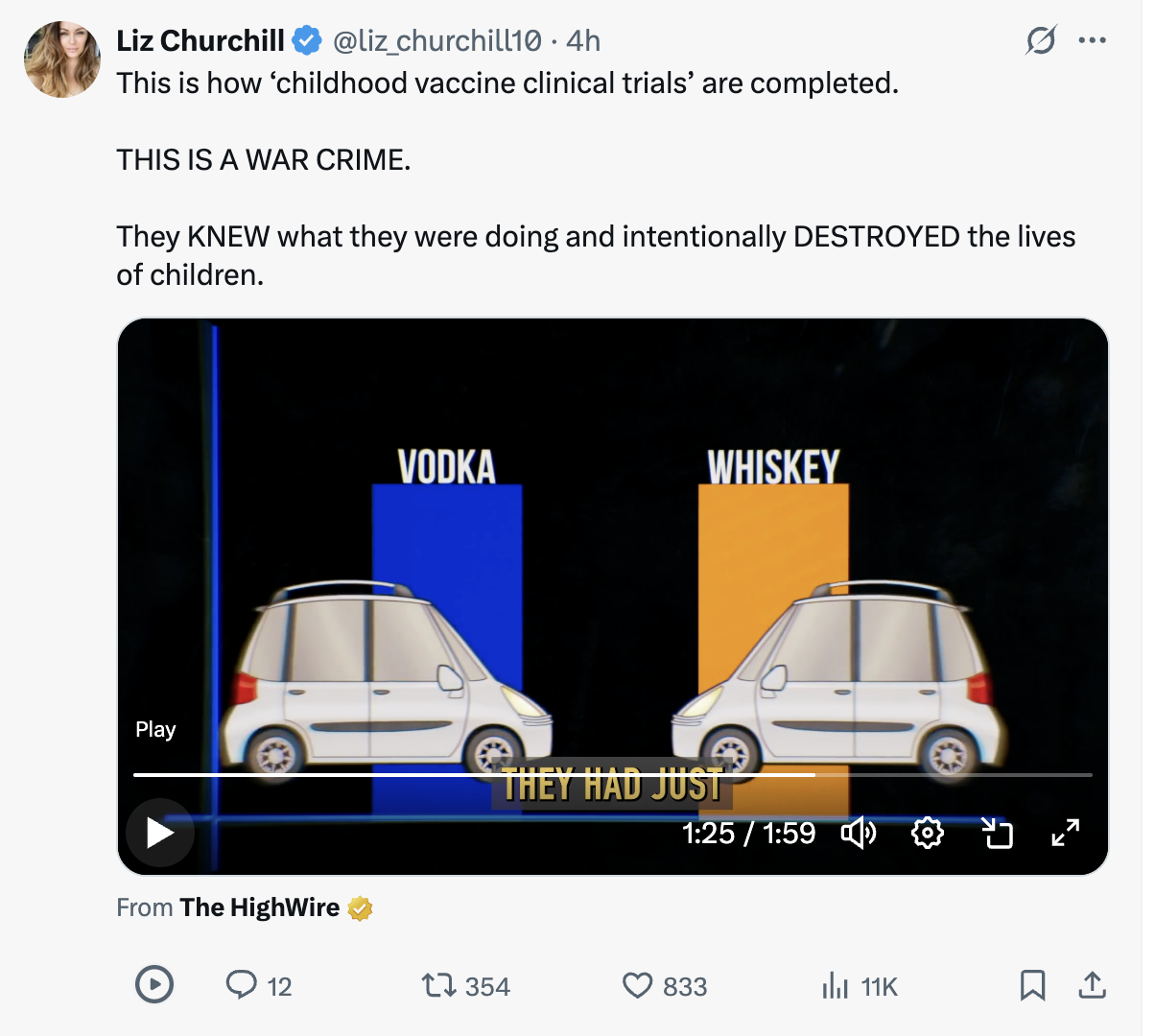

As you know, when an effective and safe vaccine already exists for a disease (e.g., measles), using an inert placebo in a clinical trial would mean deliberately denying the standard, effective treatment to the control group. A more accurate analogy than whiskey vs. vodka might be comparing a new, potentially better pain reliever to an already proven, effective pain reliever versus comparing it to a sugar pill when the patient is in severe pain.
While active controls don’t isolate all side effects caused by the vaccine ingredients compared to a perfectly inert injection, they do test if the new vaccine is significantly safer or more dangerous than the current standard of care.
Vaccine safety is always assessed in randomized, controlled clinical trials before licensure and mass use. This involves three phases:
Phase I: Small-scale safety and dosage testing, often using an inert placebo.
Phase II: Expanded safety and immune response testing.
Phase III: Large-scale efficacy and safety testing (often involving thousands to tens of thousands of participants). This is the stage where the active control argument comes into play if a standard vaccine already exists.
Monitoring Continues: Even after licensure (mass inoculation), Phase IV post-marketing surveillance systems (like the Vaccine Adverse Event Reporting System – VAERS, and the Vaccine Safety Datalink – VSD) continuously monitor for rare side effects that might not have appeared in the Phase III trial.
The scientific process involves:
1. Rigorous placebo-controlled testing for first-in-class vaccines (when no standard of care exists).
2. Rigorous controlled testing (inert or active) for all vaccines, followed by
3. Continuous post-licensure monitoring.